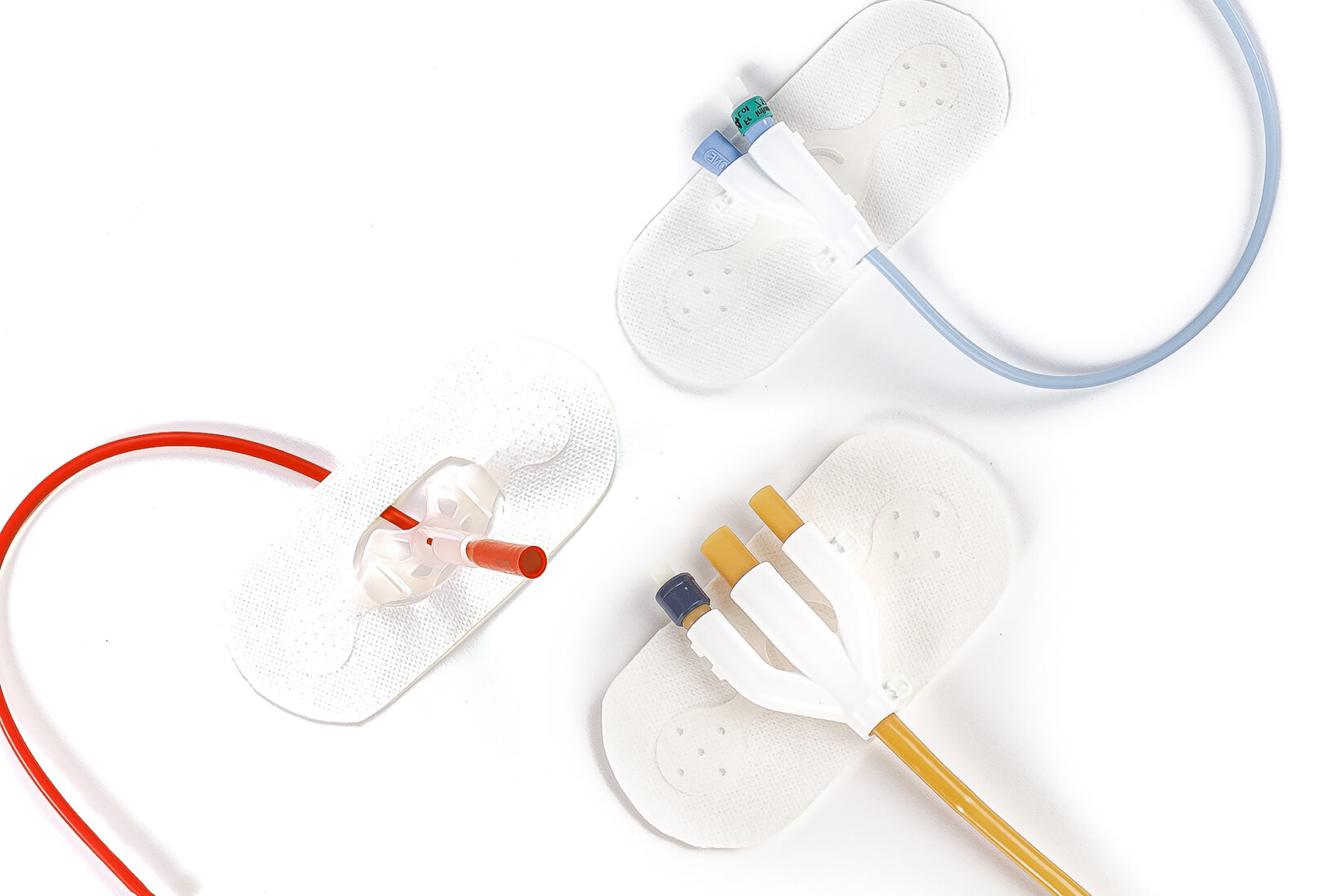
HUGO, Minn., Feb. 27, 2020 /PRNewswire/ — Levity Products Incorporated is pleased to announce the release of a set of newly patented and clinically proven external catheter stabilizers, a new medical device known as The LECS that is expected to revolutionize the catheter stabilization market. With three variations, each distinctively designed for a different type of catheter, these devices provide better alternatives for improving clinical outcomes and patient’s quality of care by offering a unique method to secure both the urine and balloon ports. In addition, Levity manufactures a one-of-its-kind suprapubic catheter stabilizer designed to drastically reduce infections, a well-recognized source of increased morbidity and economic burden in acute care services.
Indwelling urinary catheters are used in the care of more than five million patients per year. Prevalence rates range from 4% in-home care to 25% in acute care. Catheter-associated urinary tract infections account for more than 40% of all nosocomial infections and can be associated with significant complications. The LECS devices are designed to avoid many catheter-associated problems by securing the catheter and properly positioning the drainage bag.
“Levity’s research and development has resulted in the most effective and innovative set of external catheter stabilizers, including two (2) devices to secure two-way and three-way catheters, and its unique suprapubic catheter stabilizer. As a result, millions of people will benefit from this medical breakthrough,” states Sarah Olson, CEO of Levity Products, Inc.
Levity Products and its LECS medical devices will be exhibiting at the EAUN20 Trade Show in Amsterdam at the RAI exhibition and convention center on March 21-23, 2020. Visitors can find the booth at KL 4.
About Levity Products Inc.:
Levity Products is a medical device company dedicated to bettering the lives of those experiencing critical and chronic conditions, specifically in the area of urology.
For more information about either the company or the products, please contact Sarah Olson, CEO at sarah@levityproducts.com.